Abstract
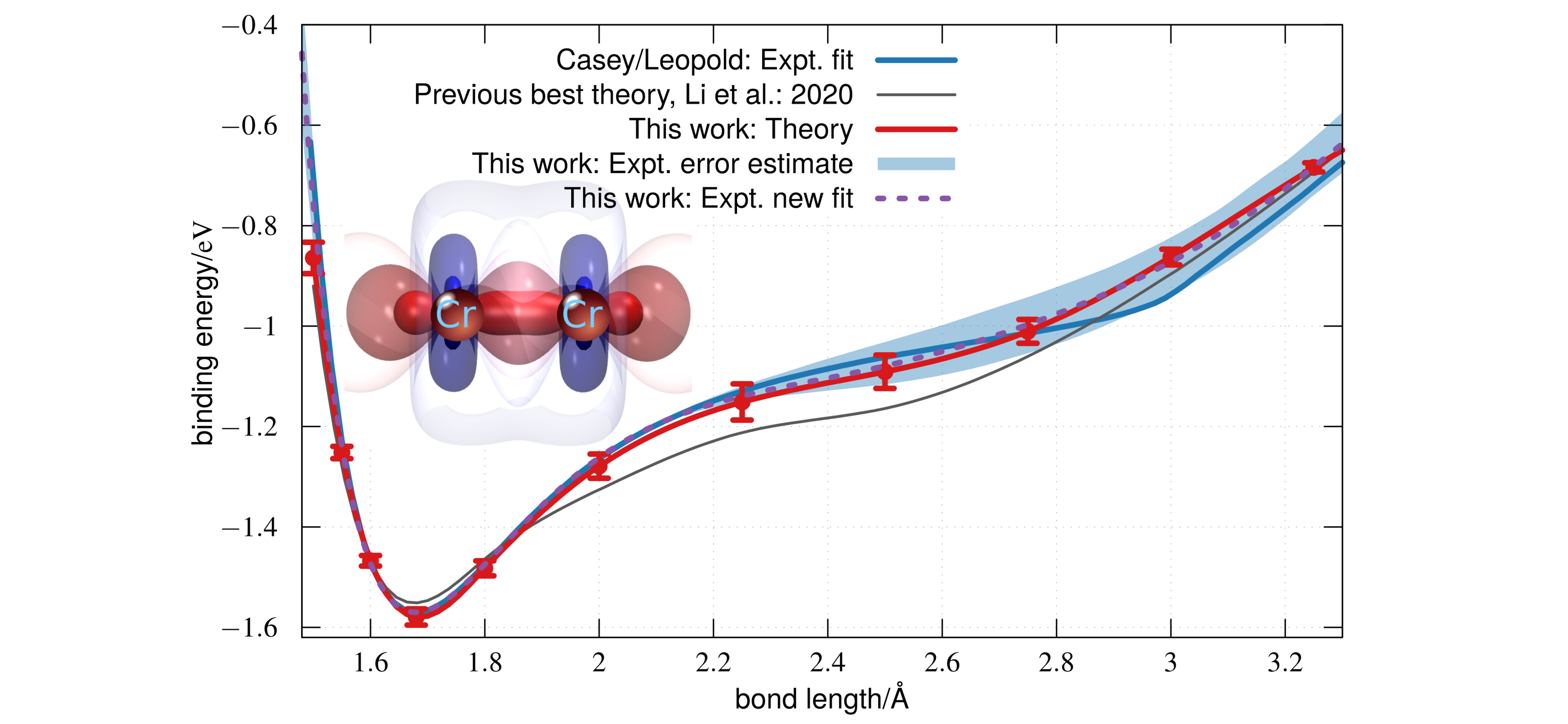
The complex electronic structure and unusual potential energy curve of the chromium dimer have fascinated scientists for decades, with agreement between theory and experiment so far elusive. Here, we present a new ab initio simulation of the potential energy curve and vibrational spectrum that significantly improves on all earlier estimates. Our data support a shift in earlier experimental assignments of a cluster of vibrational frequencies by one quantum number. The new vibrational assignment yields an experimentally derived potential energy curve in quantitative agreement with theory across all bond lengths and across all measured frequencies. By solving this long-standing problem, our results raise the possibility of quantitative quantum chemical modeling of transition metal clusters with spectroscopic accuracy.